Next Generation Animal Models for Immune System Engraftment
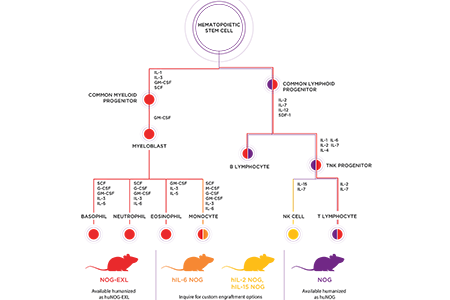
The scientists at the Central Institute for Experimental Animals (CIEA) in Japan not only developed the NOG mouse, but have been working for the past several years to generate next generation models which improve upon the state of the art in immune system engraftment. One of the problems identified in supporting proper human immune cell differentiation is that some cytokines produced by the host mouse have limited cross-reactivity to bind cognate human cytokine receptors, and consequently limit differentiation of some immune cell lineages. This problem can be overcome in a number of ways. Human cytokines can be exogenously administered or expressed via hydrodynamic plasmid injections, but that requires additional experimental manipulation of every study animal. Another approach involves transgenic expression of human cytokines. The CIEA has developed a number of such models based on the NOG mouse.Overcoming Limitations
A limitation realized early on by the field is the low number of myeloid cells in the huNOG. Several cytokines participate in myeloid cell lineage commitment, including GM-CSF and IL-3. CIEA generated a NOG mouse that expresses both human GM-CSF and human IL-3 [NOG-EXL (hGM-CSF/hIL-3 NOG)]. Upon HSC injection, this mouse supports higher overall levels of human cell engraftment as well as higher levels of myeloid cell differentiation compared to the NOG mouse. The hGM-CSF/hIL3-NOG mouse may thus be more suitable for applications requiring myeloid cells, such as allergy or immuno-oncology research related to macrophages, monocytes and their derivatives, including myeloid-derived suppressor cells (MDSCs).Researchers working with NK cells have been similarly limited by poor reconstitution of NK cells in the huNOG. Il-2 and IL-15 have been identified as important cytokines in NK cell development. CIEA generated NOG mice which express either human IL-2 or IL-15, in hopes of improving NK cell engraftment. Following human HSC engraftment, the hIL-2 NOG mice support 10 fold higher NK cell numbers compared to NOG. The hIL-15 NOG mice support expansion of human NK cells following PBMC injection.
Monocytes and macrophages are an important part of the innate immune system. Expression of human IL-6 can support monocyte differentiation or expansion, and NOG mice expressing human IL-6 (hIL-6 NOG) show enhanced cell numbers of human monocytes following HSC engraftment. These mice may be useful for immuno-oncology applications studying tumor-infiltrating macrophages.
No perfect mouse model of the human immune system exists. But researchers now have more options than ever for choosing a model that fits their scientific question.
















.jpg)

.jpg)
.jpg)
.jpg)
.jpg)





.jpg)


.jpg)
.jpg)




.jpg)





.jpg)

.jpg)