Application Areas:
NOG-EXL (hGM-CSF/hIL-3 NOG)

| Model No. | Nomenclature | Genotype |
|---|---|---|
| 13395-F | NOD.Cg-Prkdcscid Il2rgtm1Sug Tg(SV40/HTLV-IL3,CSF2)10-7Jic/JicTac | sp/sp;ko/ko;tg/wt |
| 13395-M | NOD.Cg-Prkdcscid Il2rgtm1Sug Tg(SV40/HTLV-IL3,CSF2)10-7Jic/JicTac | sp/sp;ko/y;tg/wt |
- Description
- Related Products & Services
- Price & Licensing
- Health Report
- Overview
- Genetics
- Guides & Publications
- Applications & Therapeutic Areas
- Transit, Housing & Welfare
- Diet
Overview
Nomenclature: NOD.Cg-Prkdcscid Il2rgtm1Sug Tg(SV40/HTLV-IL3,CSF2)10-7Jic/JicTac
The hGM-CSF/hIL-3 NOG is an immunodeficient CIEA NOG mouse altered to express human GM-CSF and IL-3 cytokines. The resulting transgenic mouse model supports the differentiation of human myeloid cell lineages and potentiates increased efficiency of hematopoietic stem cell and human immune system engraftment compared to the core NOG mouse.
The model was designed to express relatively low cytokine levels, which maintains the long-term stability of engraftment by preventing the exhaustion of the stem cell pool. This stability makes it a flexible platform for the generation of humanized mice.
- Supports myeloid lineage engraftment through human IL-3 and GM-CSF cytokine expression
- More efficient human hematopoietic stem cell (HSC) engraftment
- Higher levels of myeloid cell differentiation following human HSC engraftment
- Low cytokine expression and long-term HSC engraftment
Orders by weight: Taconic cannot accept orders by weight for this model. Please note that shipments may contain animals with a larger weight variation.
“Experimental Pharmacology & Oncology (EPO) GmbH uses 1st and 2nd generation NOG mice provided by Taconic for its preclinical oncology service. These mice are included in novel concepts for the development of personalized treatment options and especially suited for humanization strategies. We are very satisfied with the quality of mice, the reliability of shipment and the high level of scientific support. Further, we very much appreciate the competent and always friendly communication.”
Availability
Available now**Please note that advanced order placement is encouraged for male mice. Ordering 4 or more weeks prior to the desired shipping date is recommended when requesting males older than 3-weeks of age.
Recommended Controls
The recommended control for this model is CIEA NOG mouse.
Origin
Genetics
Guides & Publications
Initial Publication:
- Ito M, Hiramatsu H, Kobayashi K, Suzue K, Kawahata M, Hioki K, Ueyama Y, Koyanagi Y, Sugamura K, Tsuji K, Heike T, Nakahata T. (2002) NOD/SCID/γ
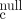 mouse: an excellent recipient mouse model for engraftment of human cells. Blood 100(9):3175-3182.
mouse: an excellent recipient mouse model for engraftment of human cells. Blood 100(9):3175-3182. - Fukuchi Y, Miyakawa Y, Kobayashi K, Kuramochi T, Shimamura K, Tamaoki N, Nomura T, Ueyama Y, Ito M. (1998) Cytokine dependent growth of human TF-1 leukemic cell line in human GM-CSF and IL-3 producing transgenic SCID mice. Leuk Res 22(9):837-43.
- Ito R, Takahashi T, Katano I, Kawai K, Kamisako T, Ogura T, Ida-Tanaka M, Suemizu H, Nunomura S, Ra C, Mori A, Aiso S, Ito M. (2013) Establishment of a human allergy model using human IL-3/GM-CSF-transgenic NOG mice. J Immunol.191(6):2890-9.
Applications & Therapeutic Areas
- Cell and Tissue Humanized
- Genetically Humanized
- Immunology
- Infectious Disease
- Inflammation
- Oncology & Immuno-Oncology
- Vaccine Research
Transit, Housing & Welfare
Need more info? Click the live chat button or Contact Us
Packing Practices
Taconic standard practice is to recombine animals of different home cages and/or ages from a single model and sex during packing, except in specific cases where Taconic's animal welfare policy prohibits recombination due to aggression or other concerns. When an order is fulfilled with animals from more than one week of birth, this standard practice results in animals from a range of birth weeks packed together in a single TTC. When an order is fulfilled with animals from genotyped models, this standard practice results in animals from different home cages packed together in a single TTC.
Customers who wish to keep animals from different weeks of birth separated should place orders with the special instruction "Divide and label by age." Note that this special request can result in increased costs for additional Taconic Transit Cages, dividers and/or freight charges.
Taconic discourages other types of custom packing requests as they can have a negative impact on animal welfare. Learn more.
Diet
- Services
- Licensing
- Pricing - USD
- Pricing - EUR
- Pricing - DKK
- Pricing - USD Nonprofit
- Pricing - EUR Nonprofit
- Pricing - DKK Nonprofit
- Select my Health Standard
- Get Custom Pricing Guide
NOG-EXL (hGM-CSF/hIL-3 NOG)
Nonprofit users (excluding users at nonprofit foundations which are affiliated with a for-profit entity): For internal research purposes, the CIEA NOG mouse® Conditions of Use for nonprofit users apply. If you wish to perform sponsored research or fee-for-service contract research using the CIEA NOG mouse®, please inquire for access conditions.
For-profit users and users at foundations which are affiliated with for-profit entities: The CIEA NOG mouse® Conditions of Use for for-profit users apply.
The CIEA NOG mouse® is produced and distributed under license rights to the following patents and trademarks:
- Japanese Patent No. 3,753,321
- US Patent No. 7,145,055; 5,464,764; 5,487,992; 5,627,059; 5,631,153; 5,789,215; 6,204,061; 6,653,113; 6,689,610
EP Patent No. 1,338,198 - Japanese Trademark Reg. No. 4,823,423
- US Trademark Reg. No. 3,118,040
- EU Trademark Reg. No. 3,736,758
Pricing - USD
Opportunist Free (OF) Health Standard
13395 Female
13395-F Genotype sp/sp;ko/ko;tg/wt
Available now
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 10 | US$394.00 |
13395 Male
13395-M Genotype sp/sp;ko/y;tg/wt
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 10 | US$276.00 |
Pricing - EUR
Opportunist Free (OF) Health Standard
13395 Female
13395-F Genotype sp/sp;ko/ko;tg/wt
Available now
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 10 | 358,00 € |
13395 Male
13395-M Genotype sp/sp;ko/y;tg/wt
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 10 | 251,00 € |
Pricing - DKK
Opportunist Free (OF) Health Standard
13395 Female
13395-F Genotype sp/sp;ko/ko;tg/wt
Available now
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 10 | kr.2.660,00 |
13395 Male
13395-M Genotype sp/sp;ko/y;tg/wt
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 10 | kr.1.860,00 |
Pricing - USD Nonprofit
Opportunist Free (OF) Health Standard
13395 Female
13395-F Genotype sp/sp;ko/ko;tg/wt
Available now
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 10 | US$189.00 |
13395 Male
13395-M Genotype sp/sp;ko/y;tg/wt
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 10 | US$169.00 |
Pricing - EUR Nonprofit
Opportunist Free (OF) Health Standard
13395 Female
13395-F Genotype sp/sp;ko/ko;tg/wt
Available now
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 10 | 173,00 € |
13395 Male
13395-M Genotype sp/sp;ko/y;tg/wt
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 10 | 154,00 € |
Pricing - DKK Nonprofit
Opportunist Free (OF) Health Standard
13395 Female
13395-F Genotype sp/sp;ko/ko;tg/wt
Available now
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 10 | kr.1.279,00 |
13395 Male
13395-M Genotype sp/sp;ko/y;tg/wt
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 10 | kr.1.145,00 |
Select my Health Standard
Need help choosing the right Taconic Biosciences health standard for your research?
Use the Health Standard Selector to enter your exclusion list. The tool will tell you which health standards meet your requirements.
Get custom pricing guide
Schedule A Scientific Consultation
Speak with a PhD-level Field Application Scientist who can help you select the most appropriate model and maximize your experimental success.
Related Models
Smart Select Program
Free Animal Model Trial: Quality & Compatibility Assurance
To help alleviate uncertainty in selecting a new model, we offer a free animal trial program that allows you to test the quality, consistency, and availability of Taconic’s rodent models. Pilot our most popular mouse or rat models in your facility at no cost to your organization with the Smart Select program.
- Evaluate a specific model strain, before making any financial investment
- Request up to 20 animals from one of the strains listed here to test for quality, consistency, and suitability for your experimental purpose
- Enjoy free ground shipping, where applicable (see table for more details)
















.jpg)

.jpg)
.jpg)
.jpg)
.jpg)





.jpg)


.jpg)
.jpg)




.jpg)





.jpg)

.jpg)














