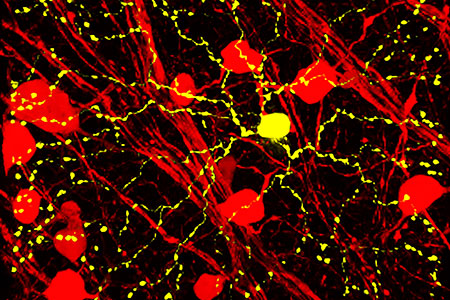
 Unique in its complexity, the central nervous system (CNS) has long represented a challenge for biomedical research, in part because the definition and characterization of constituent cell types remains incomplete. Indeed, one of the major goals behind the 2013 National Institutes of Health (NIH) launch of The Brain Research Through Advancing Innovative Neurotechnologies (BRAIN) Initiative was to "identify and provide experimental access to the different brain cell types to determine their roles in health and disease"1. This call for a unified census of all cells within the brain was strengthened and expanded in 2017 under the BRAIN Initiative Cell Census Network (BICCN). To this day, the effort aims to bring together molecular, anatomical, and functional information to provide researchers with a comprehensive understanding of the diverse cell types present in the brain and their respective function(s). Localized or limited expression of neurotransmitters or transcription factors, for example, is often used to define a cell type. And other features such as morphology and connectivity are used to help define individual cell types, though they are often difficult to leverage experimentally, and "the very definition of neuron type in many brain regions is often contentious"2. Regardless, the genetic tractability of many different model organisms (particularly the mouse) has allowed researchers a means to target and manipulate CNS cell types.
Unique in its complexity, the central nervous system (CNS) has long represented a challenge for biomedical research, in part because the definition and characterization of constituent cell types remains incomplete. Indeed, one of the major goals behind the 2013 National Institutes of Health (NIH) launch of The Brain Research Through Advancing Innovative Neurotechnologies (BRAIN) Initiative was to "identify and provide experimental access to the different brain cell types to determine their roles in health and disease"1. This call for a unified census of all cells within the brain was strengthened and expanded in 2017 under the BRAIN Initiative Cell Census Network (BICCN). To this day, the effort aims to bring together molecular, anatomical, and functional information to provide researchers with a comprehensive understanding of the diverse cell types present in the brain and their respective function(s). Localized or limited expression of neurotransmitters or transcription factors, for example, is often used to define a cell type. And other features such as morphology and connectivity are used to help define individual cell types, though they are often difficult to leverage experimentally, and "the very definition of neuron type in many brain regions is often contentious"2. Regardless, the genetic tractability of many different model organisms (particularly the mouse) has allowed researchers a means to target and manipulate CNS cell types. This experimental strategy is frequently accomplished using an approach that combines two distinct mouse models. For example, genetically engineered mice may be created which use known regulatory sequences to drive expression of a site-specific recombinase (e.g., Cre) in a specific population of cells. Once available, such a model can be crossed to one of many others that are designed for unique purposes: eliminating the expression of a gene (e.g., conditional knockout) or allowing expression of a fluorescent reporter (e.g., for lineage tracing), ion channel (e.g., for optogenetic activation), toxin (e.g., for cell type-specific ablation), or viral receptor (e.g., for transsynaptic tracing). Due to the early addition of Cre recombinase to the genetically engineered mouse toolbox, a wide variety of mouse lines now exist which can be used in this interchangeable manner — by pairing one line expressing Cre with others allowing for the desired manipulation to occur after Cre-mediated recombination. Cre-expressing mouse lines are thus often referred to as "drivers" because they will mediate recombination and drive genomic alterations as well as corresponding functional consequences.
This Insight will first present an introduction to several resources that can be used to identify, evaluate, and choose an appropriate Cre-driver — with an emphasis on those appropriate for use in CNS research. It will then highlight recent advances in model generation technology that have reduced the time and resources required to generate a new Cre-driver, as this may often represent the best way to address a specific biological question.
Neuroscience-Focused Cre-Driver Resources
Following the first demonstrations that the Cre/loxP system worked efficiently in mammalian cells, an exponential increase occurred in the generation and characterization of Cre-based mouse model resources available to the research community3,4. Ultimately, this increase extended to include the development of resources intended to allow access to different neuronal cell types — not only by individual laboratories, but also through larger and more organized efforts.One example of such an organized effort is the Gene Expression Nervous System Atlas (GENSAT) project, which originally used modified bacterial artificial chromsomes (BACs) to express enhanced green fluorescent protein (EGFP) under the control of regulatory elements for genes known or suspected to have roles in the CNS5. Following this initial effort, a similar approach was used to generate different BAC transgenic lines expressing either constitutive or inducible Cre recombinase6. Ultimately, over 250 BAC Cre-driver lines were generated and made available through the Mutant Mouse Resource & Research Centers (MMRRC GENSAT/NINDS Collection). While many of these lines have been well-characterized, these models are subject to traditional concerns usually associated with random transgenesis7. Most notably, the potential for incomplete or inaccurate expression arising from position effects and copy number variation. Additionally, many GENSAT lines are available only on a complex (i.e., STOCK) genetic background and as cryopreserved stock, which may present obstacles for their use and the interpretation of results.
Similarly, the NIH Neuroscience Blueprint Cre Driver Network generated a large number of Cre-driver lines in an effort that has spanned multiple institutions and utilized different methods for their generation (e.g., enhancer trapping, targeted mutation). Many of these Cre-driver lines are also characterized and publicly available (MMRRC Neuroscience Blueprint Collection)8. Similar to the GENSAT Cre-drivers, many of these lines were generated using a non-targeted approach or may be available only on a complex genetic background and as cryopreserved stock.
Many other Cre lines exist and are publicly available, though they often fall outside of a large-scale collection or characterization effort. Thus, a careful search of general resources — including the International Mouse Strain Resource (IMSR) and Mouse Genome Informatics (MGI) databases — should also be conducted to determine whether existing lines are of interest (and available).
The strategy of placing Cre expression under the control of promoter and/or regulatory sequences of individual genes such as transcription factors (e.g., Emx, Cux2, Ascl1, Dlx1) or neurotransmitters and neuropeptides (e.g., Chat, Pomc, Th, Dbh) has no doubt predominated. However, this strategy comes with the caveat that "single gene-driven recombinase lines often mark relatively broad and mixed cell populations" within the CNS2. Such broad expression patterns — which may include cell types beyond those being directly studied — can be artificial, resulting from the nature of the modification or transgene itself. Or they can result directly (and appropriately) from the promoter used, whether or not the complete expression profile for that gene was known and appreciated beforehand. For this reason, one should always consult primary literature and independent databases in order to understand the full extent of Cre expression observed in a specific line, and whether it deviates from expectation.
One example of such a database is the Transgenic Characterization Database, associated with the Allen Brain Atlas and first described in detail by Harris and colleagues, with their analysis of 135 Cre-driver lines across 295 separate brain structures9. Another example is the Recombinase Activity Database from MGI, which contains information on over 900 Cre-drivers assayed for expression in the CNS (>850 with expression detected). Importantly, many assessments are based on expression at a particular age or set of ages, and thus temporal changes in gene expression should be considered. If expression at a particular stage is important for your work but that timepoint has not been included, it may be best to examine this first-hand. Further, many reporters used in the process of such characterization may be permanently expressed in a given lineage, if and when Cre expression itself subsides. Thus, reporter expression at a particular timepoint or in a particular cell population may indicate prior Cre expression alone (e.g., within a particular progenitor cell population that may or may not also persist). Finally, many loxP-flanked alleles show different sensitivities to recombination. As a result, the absence of reporter expression does not guarantee a lack of Cre expression or activity, and its presence does not guarantee that a separate floxed allele (e.g., allowing for conditional knockout of a separate target gene) has also undergone recombination. Combined with the risk for unintended germline recombination, these represent several of the key risks to be aware of when using Cre-driver lines10. If and when the need for precise temporal control is present — for example, to circumvent undesirable effects or coordinate with other experimental manipulations — a Cre-driver expressing an inducible form of Cre (e.g., Cre-ER or Cre-ERT2) should also be considered.
One example that provides a contrast to cell-type-specific Cre-drivers based on anatomically restricted gene expression patterns are those that function instead on neuronal activity. Examples include drivers which do so by taking advantage of immediate early genes such as Fos or Arc11. These can be used to provide access to different neurons on the basis of their activity following a particular stimulus. Further, while Cre-drivers based on individual gene expression patterns may only allow a certain degree of specificity, using these in combination with other genetic tools (e.g., reporters) or manipulations (e.g., viral infection) that also utilize restricted expression or delivery can increase specificity through an intersectional approach12,13. Cre-drivers may also be coupled with the use of other site-specific recombinases such as Flp for intersectional targeting, and efforts to increase the availability of alternate recombinase-drivers have been described14. Given the wealth of possibilities and existing techniques, however, a thorough discussion of such methods is outside of the scope of this article.
Generation of Novel Cre-Driver Lines using CRISPR-Mediated Approaches
If an appropriate Cre-driver cannot be identified, obtained, or is not present on the appropriate genetic background, then the generation of a new Cre-driver may represent the best solution — both experimentally and practically (with respect to the time and resources invested). While different methods may be used to generate a new Cre-driver, the recent development of Easi-CRISPR technology allows for the targeted knock-in of DNA sequences of approximately 1-2 kb into endogenous loci relatively quickly and with reduced effort and cost.Easi-CRISPR utilizes a long single-stranded DNA (lssDNA) repair template (approx. 1-2 kb in length with homology arms of 50-100 bp), rather than the single-stranded oligodeoxynucleotide (ssODN) templates typically used for knock-in of smaller sequences (e.g., point mutations, epitope tags)15. This increase in repair template size allows for the knock-in of recombinase (e.g., Cre, Flp) sequences via CRISPR/Cas9 genome editing directly in zygotes. Microinjection of the lssDNA repair template along with other CRISPR/Cas9 reagents can lead to the generation of founder animals in as little as 12 weeks. Expansion from founder animals can in turn generate heterozygous offspring in the next generation, which may be used to establish a permanent colony or allow for pilot cohort production.
Apart from the relatively short timeline, Easi-CRISPR also offer other advantages when compared to traditional gene targeting performed in ES cells. First, it allows model generation on a wider range of genetic backgrounds, including many commercially available inbred or outbred strains as well as pre-existing or proprietary models that already contain separate modifications of interest. Second, Easi-CRISPR can be performed by directly delivering the associated reagents into fertilized zygotes. As with other more basic CRISPR/Cas9 methods (e.g., creation of a constitutive knockout), this does not require the use of any positive or negative selection strategy, such as the drug cassettes that are often used to help identify successfully targted ES cells. After the removal of such selection cassettes (either in vitro or through breeding), residual target sites (e.g., loxP or FRT sites) will also remain. And while it is routine to design a targeted allele such that these sites remain in an area with low sequence conservation, it is still possible that their presence may affect gene expression. As Easi-CRISPR does not require such selection cassettes, it has the potential to allow successful targeting without leaving any artificial or exogeneous sequences behind.
Traditional techniques for random transgenesis may be employed with a timeline similar to CRISPR/Cas9 gene editing in zygotes. Easi-CRISPR, however, offers advantages when contrasted with these techniques. First, Easi-CRISPR allows for targeted knock-in into specific genomic locations. When the endogenous locus for the gene-of-interest is targeted, this can allow either the simultaneous knockout of the endogenous gene or expression of Cre recombinase from a polycistronic mRNA. One recently published example of the latter comes from the generation of Slc26a9P2ACre via Easi-CRISPR16. P2A is one of several small 2A peptides (along with T2A, E2A, and F2A) that were discovered in viral genomes and are commonly used to allow for the expression of multiple proteins from a polycistronic vector. This is made possible by ribosome skipping that occurs near the C-terminal end of the 2A sequence, which leaves the majority of the 2A peptide sequence (approx. 20 amino acids) attached to the upstream protein. A single proline residue, in contrast, is left attached to the downstream protein. This method is often preferred over the use of an IRES (internal ribosome entry site) sequence due to the relatively small size of the 2A peptide and a reduced risk for lower expression of the downstream protein (often observed with IRES utilization). The use of 2A peptides may also be preferred over creation of a fusion protein, as it presents a lower risk for disruption to the expression, localization, and function of the protein-of-interest. If a 2A peptide approach is undesirable, Cre insertion can also be targeted to a safe-harbor locus and placed under the control of custom regulatory sequences. Both approaches offer more reliable and/or predictable expression when compared with random transgenesis, in which transgene expression is subject to position effects, copy number variance, and the final structure of the transgene. Second, the targeted knock-in facilitated by Easi-CRISPR allows the user to bypass the expansion and characterization of multiple founder lines that is often required for new models created through random integration transgenesis. The time and resources required for these steps are often underestimated at project start, and may prove to be a significant obstacle when creating a new transgenic model.
Constraints on the use of Easi-CRISPR currently include the upper limit of repair template length (approx. 1-2 kb), as well as a requirement for careful validation of both on- and off-target modifications. Both are strongly recommended, due to the difficulty of producing the lssDNA repair template as well as the potential for complex outcomes that can include partial repair and unexpected mutations. Continued improvement to this and other model generation technologies should, however, allow for the ongoing generation of new Cre-driver mice which can serve as tools for probing CNS biology.
Schedule Consultation
















.jpg)

.jpg)
.jpg)
.jpg)
.jpg)





.jpg)


.jpg)
.jpg)




.jpg)





.jpg)

.jpg)



