Application Areas:
hACE2 AC70 Mouse

| Model No. | Nomenclature | Genotype |
|---|---|---|
| 18222-F | B6;C3-Tg(CAG-ACE2)70Ctkt | tg/wt |
| 18222-F | B6;C3-Tg(CAG-ACE2)70Ctkt | wt/wt |
| 18222-M | B6;C3-Tg(CAG-ACE2)70Ctkt | tg/y |
| 18222-M | B6;C3-Tg(CAG-ACE2)70Ctkt | wt/y |
- Description
- Data
- Price & Licensing
- Health Report
- Overview
- Genetics
- Guides & Publications
- Applications & Therapeutic Areas
- Transit, Housing & Welfare
- Diet
Overview
Nomenclature: B6;C3-Tg(CAG-ACE2)70Ctkt
- Mice expressing human ACE2 are susceptible to infection by SARS-CoV-2, the virus that causes COVID-19. The hACE2 AC70 mouse model expresses human ACE2 cDNA under the control of the CAG promoter.
- ACE2 is the receptor bound by SARS-CoV-2 and SARS-CoV spike proteins during viral entry into host cells; these spike proteins have much higher binding affinity to human ACE2 compared to mouse ACE2.
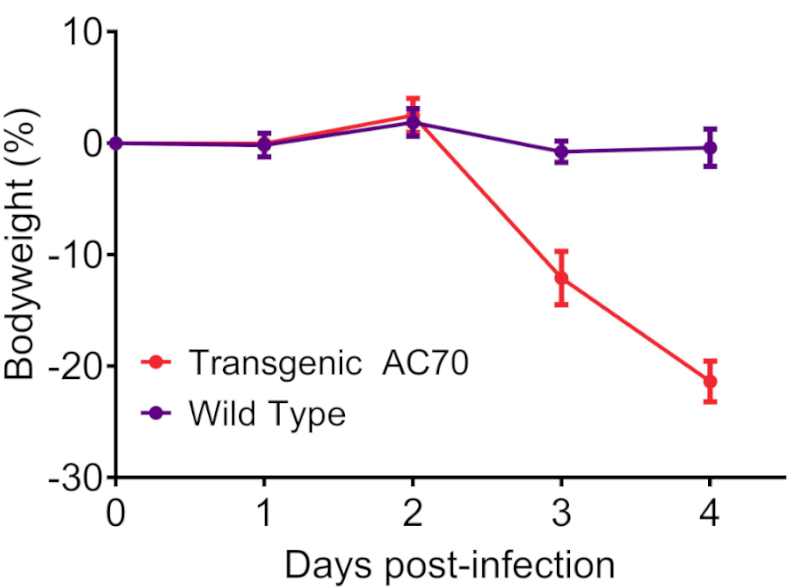
- Following intranasal infection with 106 TCID50 SARS-CoV-2, hACE2 AC70 mice lost significant bodyweight by 3 days post-infection (dpi) and 100% of mice died by 5 dpi, whereas wild type control littermates maintained bodyweight and no mice died. Personal communication, Kent Tseng, UTMB. Please see hACE2 AC22 if interested in a lethality-resistant SARS-CoV-2 infection model.
- According to published reports, hACE2 AC70 mice express human ACE2 in lungs, kidneys, liver, heart, skeletal muscle, spleen, lymph nodes, pancreas, gastrointestinal smooth muscle and ganglia, vascular endothelium, adrenal and central nervous system tissues. Taconic has confirmed hACE2 mRNA expression in hemizygous male and female mice in the following tissues: lung, brain, stomach, small intestine, cecum, colon, kidney, heart and muscle. For most tissues, mRNA expression of human ACE2 is significantly higher than that of mouse Ace2.
- Due to the susceptibility of this mouse to SARS-CoV-2, these mice should be handled in a manner consistent with appropriate guidelines for prevention of human infection with the SARS-CoV-2 virus. Proper PPE and handling methods should be used at all times. Learn more here.
- TThe hACE2 AC70 line is also susceptible to SARS-CoV, the virus which causes SARS. Following intra-nasal infection with SARS-CoV, hACE2 AC70 mice rapidly lose weight and die within days. SARS-CoV virus replicates primarily in lung and brain, and hACE2 AC70 mice develop moderate interstitial pneumonia.
- The AC70 transgene is located on the X chromosome, with one intact copy of the transgene.
- Learn more about hACE2 mice for COVID-19 research.
- Non-profit researchers may obtain breeding rights by executing a limited breeding license. This license carries a nominal fee.
| hACE2 Strain | AC70 | AC22 | Rosa26-K18 | |
| Taconic Model # | 18222 | 18225 | 18675 | |
| Nomenclature | B6;C3-Tg(CAG-ACE2)70Ctkt | B6;C3(C)-Tg(CAG-ACE2)22Ctkt | C57BL/6NTac-Gt(ROSA)26Sortm7373_A-A10(KRT18-ACE2)Tac | |
| Transgene Location | X chromosome | Unplaced scaffold region, presumably on chromosome 10 | Rosa26 locus (safe harbor) on chromosome 6, under control of the K18 promoter | |
| Transgene Copy Number | 1 | ~30–40 | 1 | |
| SARS-CoV-2 Dose (US_WA-1/2020) | 103–106 TCID50 | 101 TCID50 | 105 TCID50 | 104 PFU (or ~103.8TCID50) |
| SARS-CoV-2 Dose (Delta B.1.617.2 variant) | N/A | N/A | 104 PFU (or ~103.8TCID50) | |
| SARS-CoV-2 Dose (Omicron BA.1 variant) | N/A | N/A | 104 PFU (or ~103.8TCID50) | |
| Mortality | 100% | 100% | 30–40% | WA-1 and Delta: 25% |
| Survival (days post-infection) | 4–5 | 6–10 | 7 | 15-65 depending on viral strain/titer |
| Clinical Signs | Severe weight loss, lethal infection | Moderate–severe weight loss, lethal infection | Moderate weight loss; lethal infection in some mice | Increased systemic inflammatory response |
| Site of Viral Replication | Primarily lung and brain | Primarily lung and brain | Primarily lung and brain | Primarily lung |
| Sex Differences | Minimal sex differences observed | Some sex differences observed; males display more severe disease phenotype | ||
| Other Information | LD50: 3 TCID50 ID50: 0.5 TCID50 | ID50: 101.5 TCID50 (~30 TCID50) | LD50: 1.84 x 104 PFU | |
Orders by weight: Taconic cannot accept orders by weight for this model. Please note that shipments may contain animals with a larger weight variation.
Availability: Females readily available. 8+ week lead time for males at typical study ages.
Recommended Controls
The recommended control for this model is wild type (WT) littermates.
Origin
The hACE2 AC70 Mouse was generated by Kent Tseng et al. at the University of Texas Medical Branch at Galveston via pronuclear microinjection of a transgene into C57BL/6J x C3H/HeJ F1 zygotes. The transgene consists of human ACE2 cDNA under the control of the CAG promoter, consisting of the cytomegalovirus immediate-early enhancer and the chicken beta-actin promoter with the rabbit globin splicing and polyadenylation site. Multiple founder lines were established, including the AC70 line, which was subsequently backcrossed to C57BL/6. Taconic received the line in 2020, and the line was expanded via IVF rapid expansion using C57BL/6NTac females and hemizygous males. Taconic maintains breeding using C57BL/6NTac females by hemizygous males and/or hemizygous females by C57BL/6NTac males.
SARS-CoV-2 provokes lethal infection in hACE2 AC70 mice
Genetics
Guides & Publications
Initial Publication:
- Tseng, C.-T. K.; Huang, C.; Newman, P.; Wang, N.; Narayanan, K.; Watts, D. M.; Makino, S.; Packard, M. M.; Zaki, S. R.; Chan, T.-S.; Peters, C. J. Severe Acute Respiratory Syndrome Coronavirus Infection of Mice Transgenic for the Human Angiotensin-Converting Enzyme 2 Virus Receptor. Journal of Virology 2007, 81 (3), 1162–1173.
- Yoshikawa, N.; Yoshikawa, T.; Hill, T.; Huang, C.; Watts, D. M.; Makino, S.; Milligan, G.; Chan, T.; Peters, C. J.; Tseng, C.-T. K. Differential Virological and Immunological Outcome of Severe Acute Respiratory Syndrome Coronavirus Infection in Susceptible and Resistant Transgenic Mice Expressing Human Angiotensin-Converting Enzyme 2. Journal of Virology 2009, 83 (11), 5451–5465.
Applications & Therapeutic Areas
- Infectious Disease
- Vaccine Research
Transit, Housing & Welfare
Need more info? Click the live chat button or Contact Us
Packing Practices
Taconic standard practice is to recombine animals of different home cages and/or ages from a single model and sex during packing, except in specific cases where Taconic's animal welfare policy prohibits recombination due to aggression or other concerns. When an order is fulfilled with animals from more than one week of birth, this standard practice results in animals from a range of birth weeks packed together in a single TTC. When an order is fulfilled with animals from genotyped models, this standard practice results in animals from different home cages packed together in a single TTC.
Customers who wish to keep animals from different weeks of birth separated should place orders with the special instruction "Divide and label by age." Note that this special request can result in increased costs for additional Taconic Transit Cages, dividers and/or freight charges.
Taconic discourages other types of custom packing requests as they can have a negative impact on animal welfare. Learn more.
Diet
Data
Download document: Virulence of SARS-CoV-2 variants Washington (US_WA-1/2020), Delta (B.1.617.2), and Omicron sub-variants (BA.4/.5) in the hACE2 AC70 mouse. Data provided by Bioqual, Inc.

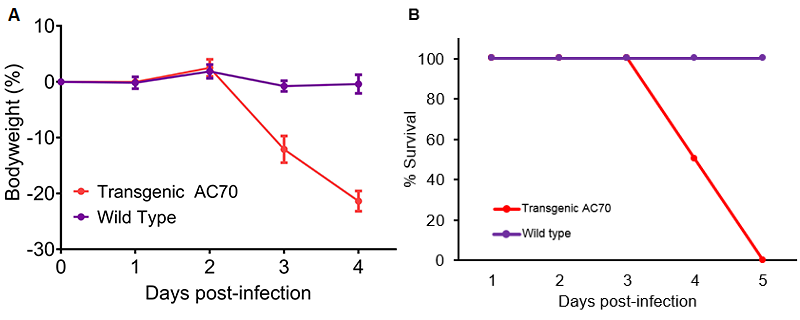
Figure 1: SARS-CoV-2 infection of human ACE2 transgenic AC70 mice resulted in an acute onset of disease and mortality. Human ACE2 transgene-positive (Transgenic AC70) and -negative (Wild type) littermates from the AC70 line were infected intranasally with 106 TCID50 of SARS-CoV-2 (US_WA-1/2020). Challenged mice were monitored daily for weight loss (% ± SEM) (A) and accumulated mortality (B). Personal communication, Kent Tseng, UTMB.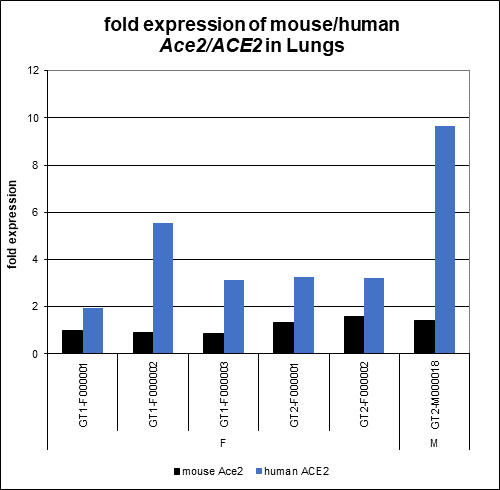
Figure 2: hACE2 AC70 mice express higher levels of human ACE2 mRNA in the lung compared to mouse Ace2 mRNA.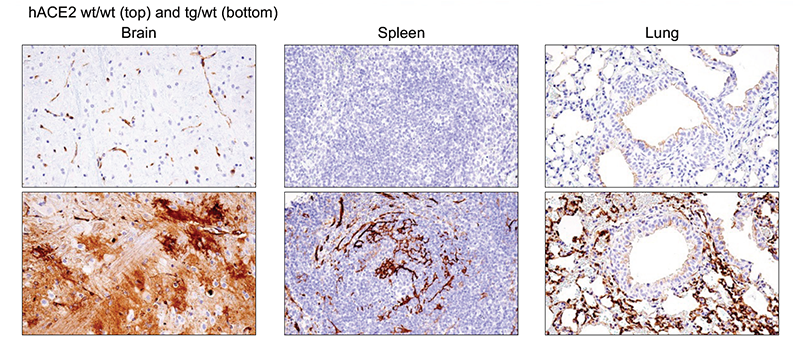
Figure 3: Immunohistochemical analysis of paraffin-embedded brain (left), spleen (middle), and lung (right) from wild type (wt/wt, top)and hACE2 AC70 (tg/wt, bottom) mice using ACE2 (E5O6J) XP® Rabbit mAb #92485 from Cell Signaling Technology, Inc.
- Licensing
- Pricing - USD
- Pricing - EUR
- Pricing - DKK
- Pricing - USD Nonprofit
- Pricing - EUR Nonprofit
- Pricing - DKK Nonprofit
- Select my Health Standard
- Get Custom Pricing Guide
Licensing
Taconic Transgenic Models™ (Models) are produced and distributed under rights to patents and intellectual property licensed from various institutions. Taconic sells the Models to purchasers, grants to each purchaser a right under Taconic's rights in such licensed patents and intellectual property to use the purchased Model in consideration of purchasers' acknowledgement of and agreement to the Terms and Conditions for Taconic Models, Products and Services and the following terms of use:
- Title to these Models and biological materials derived from them remains with Taconic.
- The Models will be used for research purposes only.
- The Models will not be bred or cross-bred except to obtain embryos or fetuses required for research purposes unless additional rights have been granted in writing by Taconic.
- The Models and biological materials derived from them will not be distributed to third parties or used for commercial purposes.
- Non-profit purchasers may not use this Model and/or biological materials derived from it in sponsored research or contract research studies unless it is purchased at the for-profit price.
Pricing - USD
Opportunist Free (OF) Health Standard
18222 Female
18222-F Genotype tg/wt
Pilot-sized cohorts are readily available. Large cohort requests have a minimum 8-week lead time. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | US$175.00 |
18222-F Genotype wt/wt
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | US$140.00 |
18222 Male
18222-M Genotype tg/y
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | US$175.00 |
18222-M Genotype wt/y
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | US$140.00 |
Pricing - EUR
Opportunist Free (OF) Health Standard
18222 Female
18222-F Genotype tg/wt
Pilot-sized cohorts are readily available. Large cohort requests have a minimum 8-week lead time. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | 166,00 € |
18222-F Genotype wt/wt
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | 133,00 € |
18222 Male
18222-M Genotype tg/y
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | 166,00 € |
18222-M Genotype wt/y
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | 133,00 € |
Pricing - DKK
Opportunist Free (OF) Health Standard
18222 Female
18222-F Genotype tg/wt
Pilot-sized cohorts are readily available. Large cohort requests have a minimum 8-week lead time. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | kr.1.233,00 |
18222-F Genotype wt/wt
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | kr.986,00 |
18222 Male
18222-M Genotype tg/y
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | kr.1.233,00 |
18222-M Genotype wt/y
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | kr.986,00 |
Pricing - USD Nonprofit
Opportunist Free (OF) Health Standard
18222 Female
18222-F Genotype tg/wt
Pilot-sized cohorts are readily available. Large cohort requests have a minimum 8-week lead time. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | US$143.00 |
18222-F Genotype wt/wt
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | US$114.00 |
18222 Male
18222-M Genotype tg/y
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | US$143.00 |
18222-M Genotype wt/y
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | US$114.00 |
Pricing - EUR Nonprofit
Opportunist Free (OF) Health Standard
18222 Female
18222-F Genotype tg/wt
Pilot-sized cohorts are readily available. Large cohort requests have a minimum 8-week lead time. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | 135,00 € |
18222-F Genotype wt/wt
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | 108,00 € |
18222 Male
18222-M Genotype tg/y
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | 135,00 € |
18222-M Genotype wt/y
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | 108,00 € |
Pricing - DKK Nonprofit
Opportunist Free (OF) Health Standard
18222 Female
18222-F Genotype tg/wt
Pilot-sized cohorts are readily available. Large cohort requests have a minimum 8-week lead time. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | kr.1.008,00 |
18222-F Genotype wt/wt
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | kr.803,00 |
18222 Male
18222-M Genotype tg/y
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | kr.1.008,00 |
18222-M Genotype wt/y
Cohorts are reserved upon order placement and will take 4-8 weeks to fulfill. An estimated lead time will be provided to you within 2-3 business days.
| Age in Weeks | Quantity 1 - 999 |
|---|---|
| 4 to 12 | kr.803,00 |
Select my Health Standard
Need help choosing the right Taconic Biosciences health standard for your research?
Use the Health Standard Selector to enter your exclusion list. The tool will tell you which health standards meet your requirements.
Get custom pricing guide
Schedule A Scientific Consultation
Connect directly with a member of our Scientific Solutions team who can help you select the most appropriate model and maximize your experimental success.
















.jpg)

.jpg)
.jpg)
.jpg)
.jpg)





.jpg)


.jpg)
.jpg)




.jpg)





.jpg)

.jpg)










