 Dr. Paula Roesch recently presented a webinar on microbiological monitoring of germ-free and gnotobiotic colonies. Taconic Biosciences has decades of experience in this area and Dr. Roesch presented a thorough look at all aspects of the process, starting from tools and approaches to process validation and monitoring and moving into isolator monitoring techniques.
Dr. Paula Roesch recently presented a webinar on microbiological monitoring of germ-free and gnotobiotic colonies. Taconic Biosciences has decades of experience in this area and Dr. Roesch presented a thorough look at all aspects of the process, starting from tools and approaches to process validation and monitoring and moving into isolator monitoring techniques. She even included lessons learned from a case study where things didn't go as expected.
We present the full webinar Q&A here:
Sterilization and Validations
Please comment on the use of hydrogen peroxide as a cold sterilant.
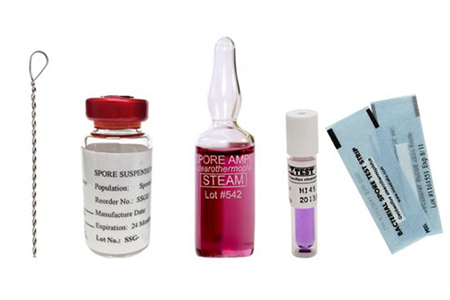
Q: Have you tried validating isolator sterilants via spore discs within the isolator?
Q: What type of sterilization approach would you take for heat-sensitive materials, such as live mammalian cells, entering an isolator?
That's the best you can do and it should work.
Water Supply Sterilization
Please discuss entry of water bottles into isolators using overnight sterilization.
We spray it thoroughly to make sure it's really soaked. We close up the port and it sits there overnight so it has a maximum contact time.
Have you ever tried irradiated water for mice?
Contaminants
What is the most common contaminant found in isolators?
16S PCR Screening for Contaminants
Have you ever had a 16S PCR positive and not found anything via culture?
Do you recommend using 16S PCR as part of a primary screening regimen or to confirm and/or identify a culture positive result?
In particular, some fastidious anaerobes are tricky. Doing the 16S helps you know that you are being as stringent as possible. It's also good to confirm a finding.
Can you offer any thoughts on the use of qPCR versus endpoint PCR for 16S detection?
The shape of the curve is actually very helpful to see. It will help you determine if it's background contamination versus a real contaminant. The PCR we do with IDEXX has a larger PCR product than a standard qPCR. It's almost 200 bases, compared to 100 bases for most assays. This also helps because the DNA fragments from feed and bedding are very small, so a larger fragment helps ensure that positive results are real.
Culture Monitoring
Have you ever used Rodac plates to monitor contamination?
I do believe they are a little more expensive than TSA with blood, but they are very effective. We just don't use them routinely for gnotobiotic monitoring; we use them more for other types of environmental monitoring.
For researchers who do not have access to an in-house microbiology lab, what recommendations do you have for how to accomplish microbiological monitoring?
We've worked with them extensively over the years. I asked them to set up the 16S PCR specifically for us. They are great to work with.
















.jpg)

.jpg)
.jpg)
.jpg)
.jpg)





.jpg)


.jpg)
.jpg)




.jpg)





.jpg)

.jpg)



